Cell Biology of Tissue Morphogenesis
Caren Norden
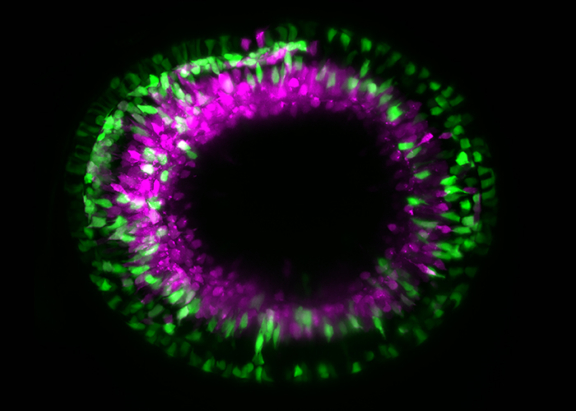
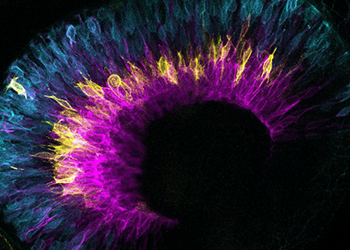
The Cell Biology of Tissue Morphogenesis lab aims to untangle the events that lead to the development of organs. In this context, the lab studies the formation of the vertebrate retina from cells to tissue and take the interactions between scales into account. The research group further assesses how mechanics influence tissue formation. Importantly, only when developmental programs occur coordinately from one stage to the next can tissues form correctly in time and space.
Thus, the group works on three key steps of retinal formation and investigates their interplay:
1. Elucidating how cells of the optic vesicle rearrange to efficiently form the neuroepithelium and the retinal pigment epithelium that together establish the optic cup.
2. Investigating the proliferation dynamics of neuroepithelial cells leading to optic cup growth until the correct number of cells is generated that poises the system for differentiation.
3. Resolving how and when neuroepithelial cells enter neurogenesis programs. Furthermore, as neurons are frequently born far away from the position at which they later function, investigating how newborn neurons move to the correct layer within the developing tissue, a process termed neuronal lamination.
To get insights into these fundamental questions, the lab combines methods of cell and developmental biology with advanced quantitative imaging, image analysis tools and, in collaboration, theoretical modeling.
Funding Member


Publications
- Karen G. Soans, Ana Patricia Ramos, Jaydeep Sidhaye, Abhijeet Krishna, Anastasia Solomatina, Karl B. Hoffmann, Raimund Schlüßler, Jochen Guck, Ivo F. Sbalzarini, Carl D. Modes and Caren Norden. (2022) Collective cell migration during optic cup formation features changing cell-matrix interactions linked to matrix topology. Current Biology
- Rana Amini, Archit Bhatnagar, Raimund Schlüßler, Stephanie Möllmert, Jochen Guck and Caren Norden (2022) Amoeboid-like migration ensures correct horizontal cell layer formation in the developing vertebrate retina. eLife 11:e76408
- Elisa Nerli, Mauricio Rocha and Caren Norden (2020) Asymmetric neurogenic commitment of retinal progenitors involves Notch through the endocytic pathway. eLife 9