Repulsive interactions bring rapid mitotic divisions to order
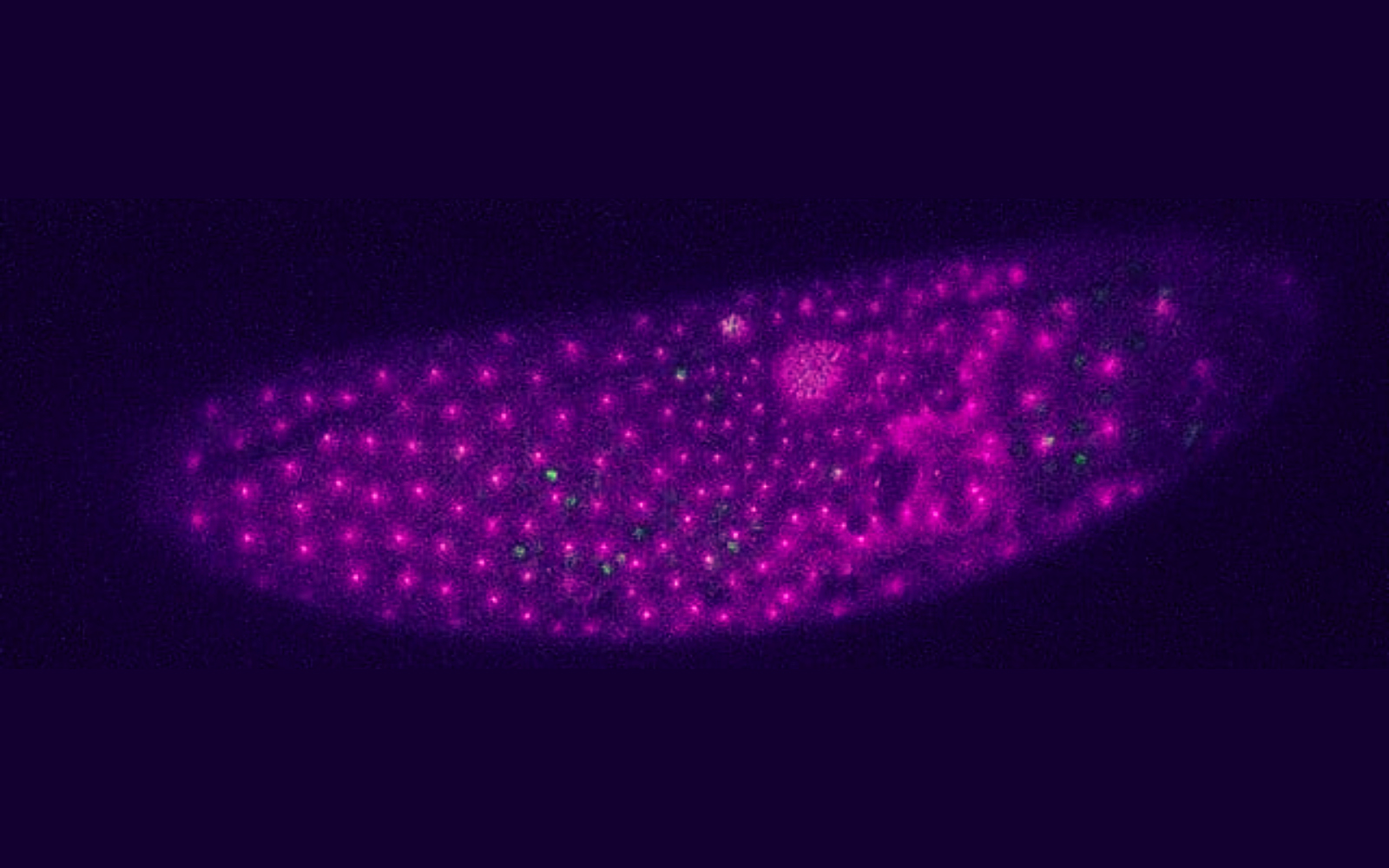
A fertilized egg cell is a common starting point for most animals. Generally, duplication of the genetic material in this cell marks the start of several rounds of division that eventually give rise to a multicellular organism. But insect species achieve this in a peculiar way: in contrast with vertebrates, they create hundreds of nuclei, each holding a copy of their genetic material, within the fertilized egg before forming individual cells.
For embryonic cells to have the same size, which is crucial for the following stages of development, nuclei need to be evenly spaced prior to being engulfed with membranes. But how exactly does the embryo achieve this order in such a chaotic setting, where hundreds of nuclei divide simultaneously? Cell and developmental biologists have studied this for decades, proposing models for the positioning of nuclei in several organisms, including frogs and mice. However, these animals produce uninuclear cells in initial stages of development, unlike insects.
A new study published in Development provides evidence for the biophysical principles that maintain the order of nuclei in fruit fly embryos. The team found that microtubules, protein polymers that are part of the cell skeleton, form a scaffold generating repulsive forces that keep nuclei at a distance. The positioning of nuclei inside multinucleated cells is very complex, encompassing a large array of possible interactions between neighboring nuclei. This type of cells develops “most robustly by an internuclear repulsion mechanism – which we present in our paper”, explains Ivo Telley, principal investigator at Instituto Gulbenkian de Ciência (IGC) and leader of the team that conducted the study. Besides maintaining nuclear positions, repulsive interactions can also reorient nuclear divisions towards unoccupied regions, where repulsion is weakest, avoiding an uneven distribution of nuclei inside large cells.
“Repulsion between nuclei was a hypothesis proposed more than twenty years ago but had never been supported by experiments”, Ivo remarks. This quest for one of “nature’s most basic principles” started in 2013 and involved collaboration with research institutes in Singapore and Germany. Soon after Ivo set up his lab at the IGC, Jorge Carvalho, first author of the study, joined him to implement the innovative experimental approach that would lead to these findings. Fruit fly embryo explants, made by extracting cytoplasm and nuclei from the embryo, allowed the researchers to unveil the mechanisms behind nuclei distribution without the need to use living samples, which are often complex and difficult to analyze. “This approach was key to most of the experimental data in the paper”, Jorge emphasized.
Ultimately, this long journey led the researchers to discover a mechanism that directly impacts cell size and positioning, two properties that define core cellular functions in later embryonic development. But these findings go beyond the development of insects: they also improve our understanding of the evolution of species, such as our own. “Internuclear repulsion was key for the transition from unicellular organisms to multicellularity”, Ivo highlights.
This study was developed at Instituto Gulbenkian de Ciência in collaboration with the National University of Singapore and the European Molecular Biology Laboratory and was supported by the Human Frontiers Science Program.
Read Paper
