Cells read the room before migrating to form tissues
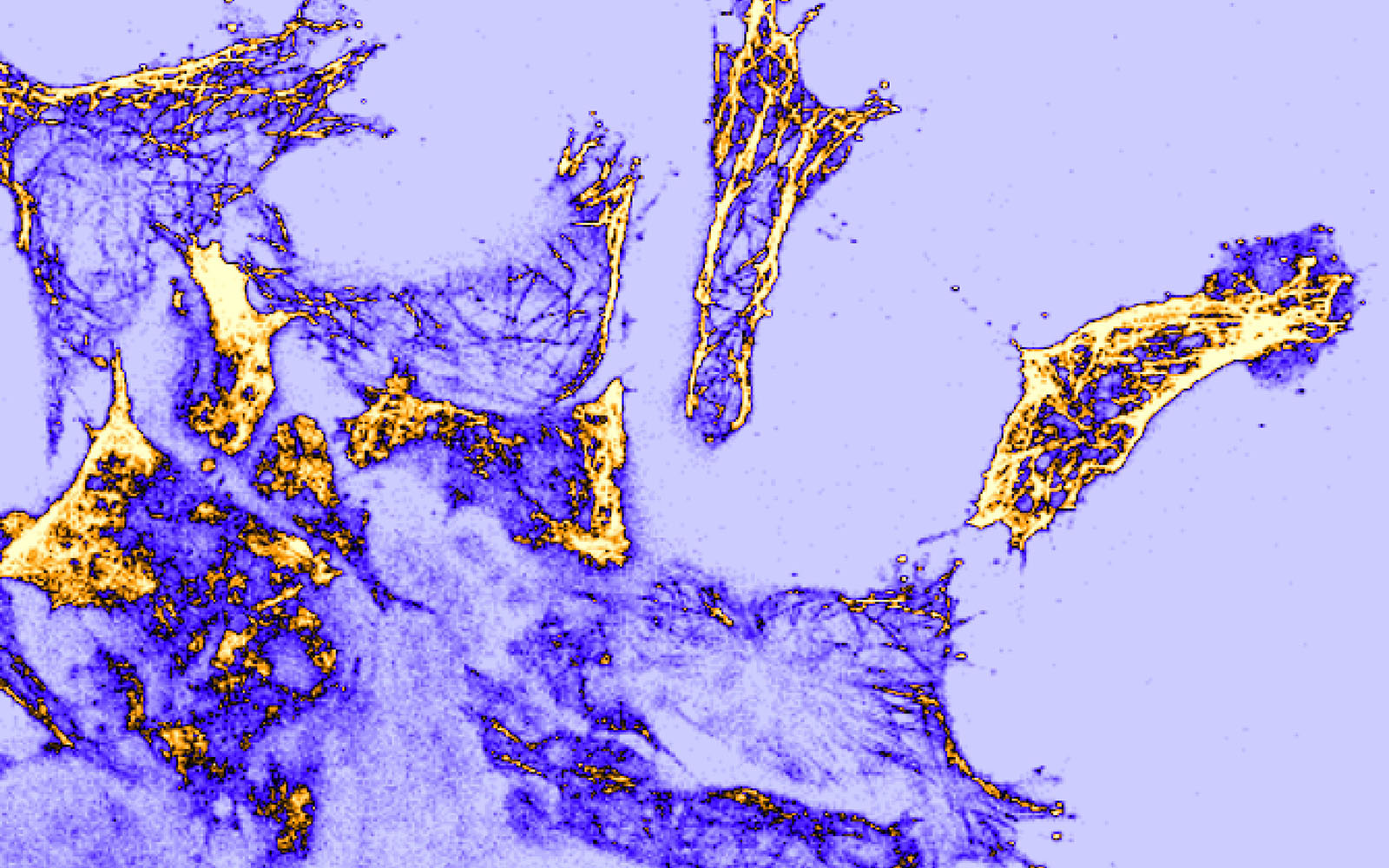
The environment within a developing embryo is very dynamic, with cells traveling coordinately to get to places where they will perform specific functions. Moving alone or in clusters, through a process known as collective cell migration, cells eventually reach their destination to form the different tissues and organs of the body. But what exactly triggers and directs the motion of multiple cells in such a coordinated way?
To migrate, cells rely on chemical and physical inputs from the surface they use as a substrate. Studies performed in Petri dishes have suggested that stiffer surfaces favor cell migration, while softer ones do not allow it. Thus, it remains unclear whether and how cells adjust their mechanical properties to their surface when migrating in the dynamic and complex landscape of a living organism.
To address this, researchers from Elias Barriga’s lab at the Instituto Gulbenkian de Ciência (IGC) studied the interplay between cells that form several structures of the head and face and their native substrate in frog embryos. By resorting to a special type of microscope (Atomic Force Microscope), the researchers were able to measure the stiffness of cells and their respective substrates in migratory and non-migratory stages.
Before these experiments, scientists thought that migrating cells matched their elastic properties to those of the surface they were laying on. But this study, published in Nature Materials, showed that, instead, cells became softer upon the stiffening of their migratory surface.
To gain further insight on the impact of cell stiffness on migration, the authors integrated the obtained data into a three-dimensional model developed by the group of Abdul Malmi-Kakkada at Augusta University, USA. Combining this model with biophysical experiments, the authors discovered that clusters of cells spread more effectively when the substrate-to-cell stiffness ratio is higher. This means that if cells within a cluster are significantly stiffer than their substrate they should fail to migrate. To travel effectively, they are required to reduce their stiffness first. Then how exactly do these cells adjust their properties to move within the embryo?
The researchers found out that cells sense the stiffness of their environment through a specific channel in their membrane, named Piezo1. This, in turn, triggers the modification of the cells’ skeleton, allowing them to tune their mechanical properties and initiate collective migration in the embryo. A surprising result arising in this study, is that the modification of this protein, and the consequent decrease in cell stiffness is sufficient for cells to migrate regardless of whether the substrate is soft of not. “This is relevant as stiff substrates were typically described as permissive surfaces for migration. In other words, scientists assumed that when the migratory substrate of cells evolved from soft to stiff, the cells would pass from walking in snow to run into pavement”, explains Elias Barriga. “Instead, our data shows that the substrate is rather informative by telling cells that they should lower their stiffness to migrate. As shown in our study, once a cell cluster reached low stiffness values, they could migrate regardless the stiffness of their substrate”, he adds.
These insights could impact our understanding of several contexts in which collective cell migration is involved. Apart from embryo development, this type of cell motion also occurs when tissues are repaired or when cancer cells undergo metastasis. Exploring these new observations could help scientists understand the migration of cancer cells, which often happens in soft tissues.
Read Paper
