SARS-CoV-2 vaccine effectiveness study in health professionals
The data on the first 1000 health professionals, vaccinated on December 2020, reveals that 90% developed antibodies in the three weeks following the administration of the first dose. The study, carried out by Instituto Gulbenkian de Ciência (IGC) and Centro Hospitalar de Lisboa Ocidental, E.P.E. (CHLO), intends to evaluate the effectiveness of the vaccine and the response to different variants, in a range of 1200 health professionals, vaccinated with the first vaccine available nationwide. This data collection has been recommended by the World Health Organization to monitor the introduction of the new vaccine internationally.
The challenges posed by the new SARS-CoV-2 virus have strengthened efforts to find mechanisms to contain and prevent the disease. The approval of the different vaccines by the supervisory authorities and the launch of the vaccination campaign marked an important milestone for controlling the epidemic. However, monitoring the vaccine’s effectiveness and impact is crucial to know the immediate and long-term effects, as many questions remain unanswered in clinical trials.
Instituto Gulbenkian de Ciência along with Centro Hospitalar Lisboa Ocidental, E.P.E. has been developing a set of surveillance projects for the SARS-CoV-2 virus in health professionals since the spring of 2020, which includes the vaccine effectiveness study (conducted on doctors, nurses, operational assistants and diagnostic and therapeutic technicians). The first results of this study, after the administration of the Pfizer / BioNTech vaccine, reveal that 90% of these health professionals already demonstrate an immune response to the vaccine. All participants were tested for antibodies to the virus before taking the vaccine and three weeks after the first dose. In a second stage, the same professionals will be tested three weeks after taking the second dose. The duration of the immune response will be monitored in subsequent stages scheduled throughout the year. Throughout the process, participants answer a questionnaire to register symptoms to assess their protection against COVID19.
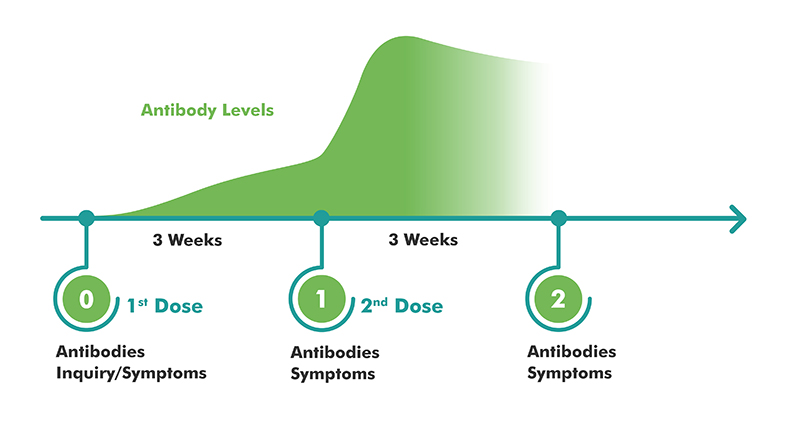
Methodology of the study of the SARS-CoV-2 vaccine effectiveness in health professionals. Curve showing the level of antibodies developed during the various stages of the study © IGC 2021
“It is necessary to monitor the application of the vaccine, in different contexts and different populations, to guarantee what scientists call “evidence in the real world ”. Only in this way can we assess data on its effectiveness in protecting against infection and disease” says Carlos Penha-Gonçalves, a Gulbenkian researcher involved in implementing the study. Jocelyne Demengeot, also a researcher at Gulbenkian, guarantees that “the data we obtained prove that the vaccine is fulfilling its first objective: to stimulate the immune system resulting in the development of virus-specific antibodies.” Monitoring of these immune responses will include the ability to neutralize different variants of the virus. From a hospital perspective, Rita Perez, Chairman of the Board of Directors of Centro Hospitalar Lisboa Ocidental, E.P.E. reinforces that “vaccines are safe and adherence has been extraordinary. It is important to monitor its introduction to ensure that it fulfils its objective of protecting those on the front lines and that it is a strong mechanism for containing the pandemic ”.
Gulbenkian also intends to extend this study by monitoring other age groups of the population, and different vaccines when available nationwide, in partnership with other hospitals and municipalities. The set of data collected will be shared with the Instituto Nacional de Saúde Dr. Ricardo Jorge, which will speed up, with the European Agencies (ECDC), the extended national monitoring that will allow to provide information for possible updates of the global policy recommendations.
Read Press Release