How large cells keep their nuclei at a distance
The inside of a cell is far from static. Dense networks of proteins and cellular structures are in constant movement, responding to their surrounding environment. The nucleus, holder of the majority of the cell’s genetic material, also moves. Inadequate positioning of this organelle can compromise core cellular functions and embryo development. When cells have only one nucleus, the cellular skeleton connects it to the inner part of the cellular membrane and positions it inside the cell. But, as is common to plants and some invertebrates, cells can have many nuclei and be extremely large, with membranes too far apart for the skeleton to work by itself. In these cases, cells need different strategies to position nuclei correctly.
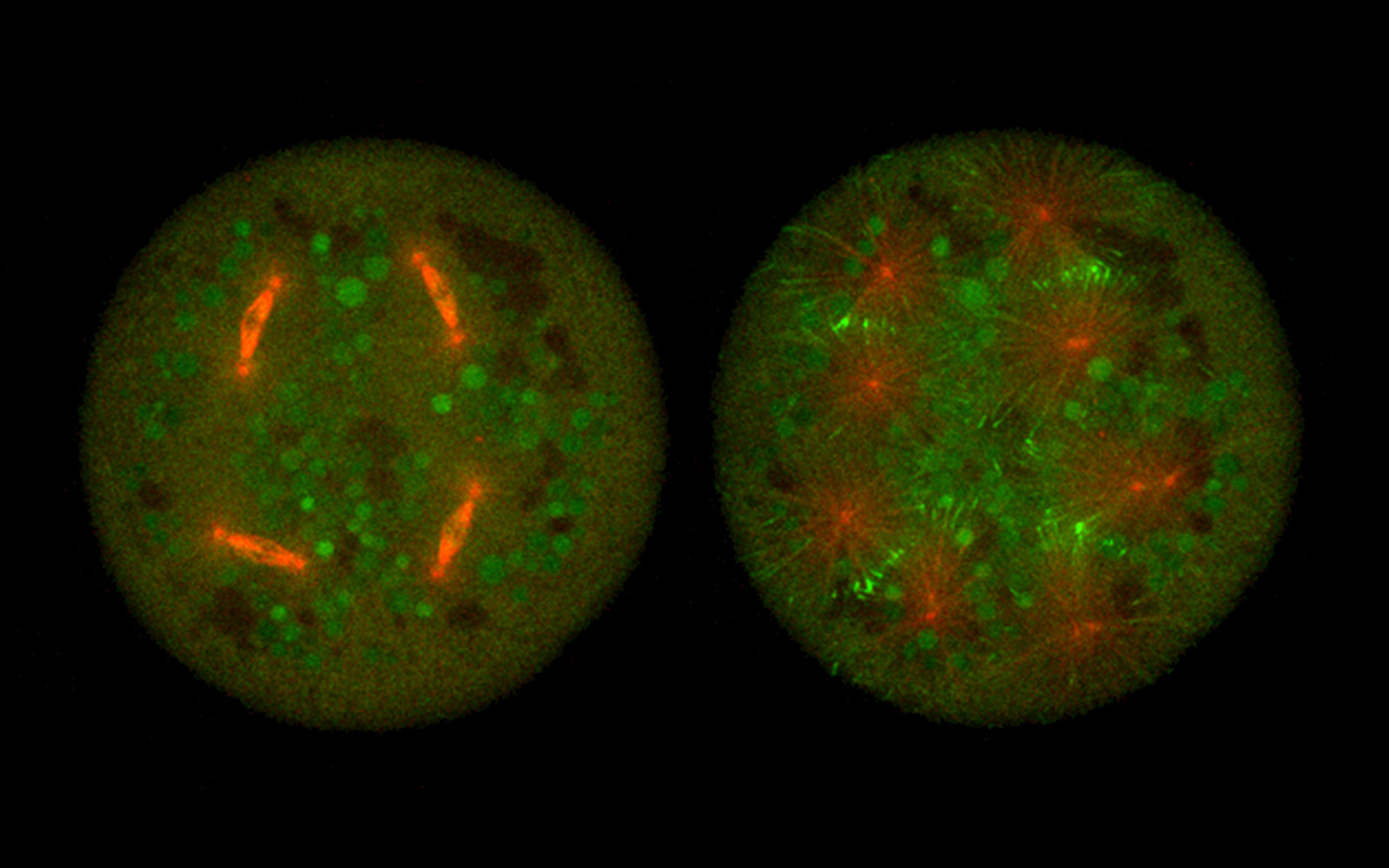
The eggs of fruit flies are an example of a single cell that is large and has many nuclei inside them. After fertilization, the egg undergoes intense nuclei proliferation, producing hundreds of these organelles, only to later form membranes around each one and individualize cells. During the process, the egg needs to disseminate positional information between nuclei and ensure they are all in the correct position. Adequate number and proper positioning of the nuclei are what determines cell size and the precision of developmental processes. But how does the cell safeguard the separation of nuclei and prevent them from colliding as their number increases?
A new study published in the Journal of Cell Biology provides evidence for two proteins required for keeping nuclei apart in the multinucleated embryo cell of the fruit fly. “When a fruit fly embryo makes cells to grow the animal, these cells need to be of the same size, from which it grows different organs. We now found that crosslinked microtubules, a component of the cell skeleton, make a stiff connection and mediate mechanical repulsion between the nuclei during the process. If nuclei are not evenly distributed, the embryo will not form cells of equivalent size, and that is detrimental for later development”, explains Ivo Telley, principal investigator at IGC and leader of the team that conducted the study.
With an approach across scientific disciplines, encompassing engineering, genetics, molecular biology, biochemistry and light microscopy, the team found that decreasing the amount of these two proteins, one that works as a crosslinker and the other as a stabilizer, leads to irregular distances between nuclei, which sometimes cluster or are missing. “Our first test experiments were immediately promising, and we were then able to show that these proteins are required for correct nuclear positioning. We also used protein biochemistry approaches to purify a deficient crosslinker that has the ability to make the internuclear connection ‘softer’ and injected it into the embryo or explants”, highlights Ojas Deshpande, first author of the study. Embryo explants were a key technical novelty to visualize this phenomenon. They were made by extracting cytoplasm and nuclei from the embryo and placing them on a surface where they can be easily accessed and manipulated. “This approach allowed us to manipulate nuclei directly, for example change their relative position”, Ivo says.
Nuclei that divide all at the same time will inevitably produce colliding trajectories. Controlling nuclear proximity enables the embryo to divide hundreds of nuclei synchronously and distribute them to any unoccupied position. This study now demonstrates a molecular mechanism that responds to short internuclear distances with repulsion forces. “The next question is what is the mechanics that keeps nuclei apart at a precise distance. In our article we discuss how microtubules and the associated crosslinker and stabilizer can define a length scale, but this remains to be tested directly, possibly with in vitro biochemical approaches. How molecules define a distance metric that is relevant for the cell is an interesting field of research”, Ivo remarks.
This study was developed at the Instituto Gulbenkian de Ciência and was supported by an EU FP7 Marie Curie CIG, an FCT PhD fellowship SFRH/BD/52174/2013, a Human Frontiers Science Program YIG and IGC core funding.
Read Paper
